Laboratory head

Student research projects



Meet the team
Research staff
-

-

-

-

-

-

About the Cardiac Cellular Systems laboratory
Heart failure is a leading cause of death worldwide. It results from a wide range of causes and is characterised by harmful cardiac remodelling and declining heart function, ultimately leading to organ failure and death.
Currently, there are no effective cures for heart failure, and fundamental questions remain unanswered about how cardiac remodelling occurs and whether it can be reversed.
Discovering the heart's cellular ecosystem
Until recently, the cellular composition of the heart was poorly understood. Using advanced genetic, flow cytometric and single-cell transcriptomic approaches, our laboratory has shed new light on what the heart is actually made of. We've demonstrated that the heart comprises a complex and diverse ecosystem of non-myocytes — the cells that support and surround heart muscle cells (Figure 1: Skelly et al. 2018, Pinto et al. 2016, Pinto et al. 2012).
These discoveries have opened new avenues of cardiovascular research, making it possible to target and manipulate specific cell types and networks. Yet virtually nothing is known about how this ecosystem of non-myocytes operates as a cell network — and that's where our research comes in.
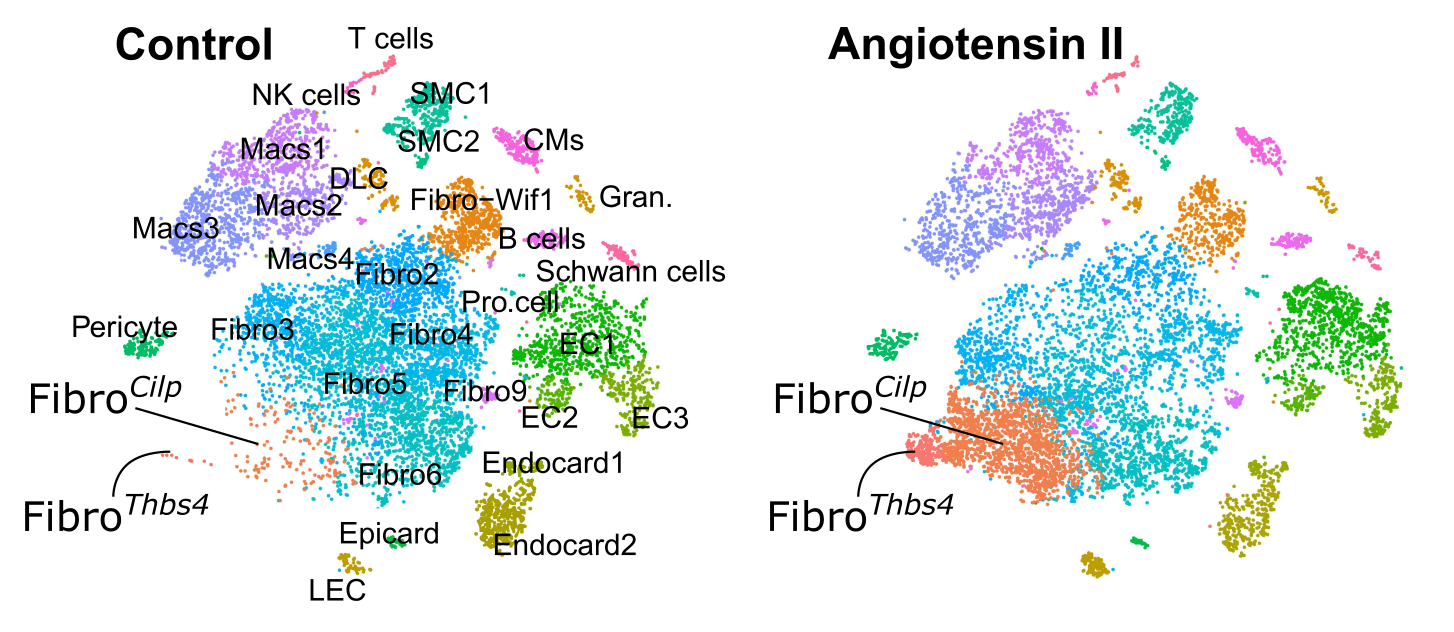
Figure 1: Understanding cardiac cellular diversity and phenotypes in tissue homeostasis and stress. The figure shows tSNE plots with dots represent individual cells. Fibro-Cilp and Fibro-Thbs4 that emerge after angiotensin II-induced fibrosis are indicated from McLellan et al. 2020.
Our research aims
We have two overarching goals:
- Understand how cardiac cell networks contribute to heart failure
We're determining how pathological changes in cardiac cell networks drive the development of heart failure. - Explore whether manipulating cell networks can prevent or treat heart disease
We're investigating whether influencing the cell network can alter cardiac remodelling and be used to prevent or treat cardiac pathologies (Figure 2).
Using single-cell technologies, data science, micro-anatomy and traditional mouse genetics, our research takes an integrative systems biology approach to uncovering the fundamental processes governing cell networks in the heart.
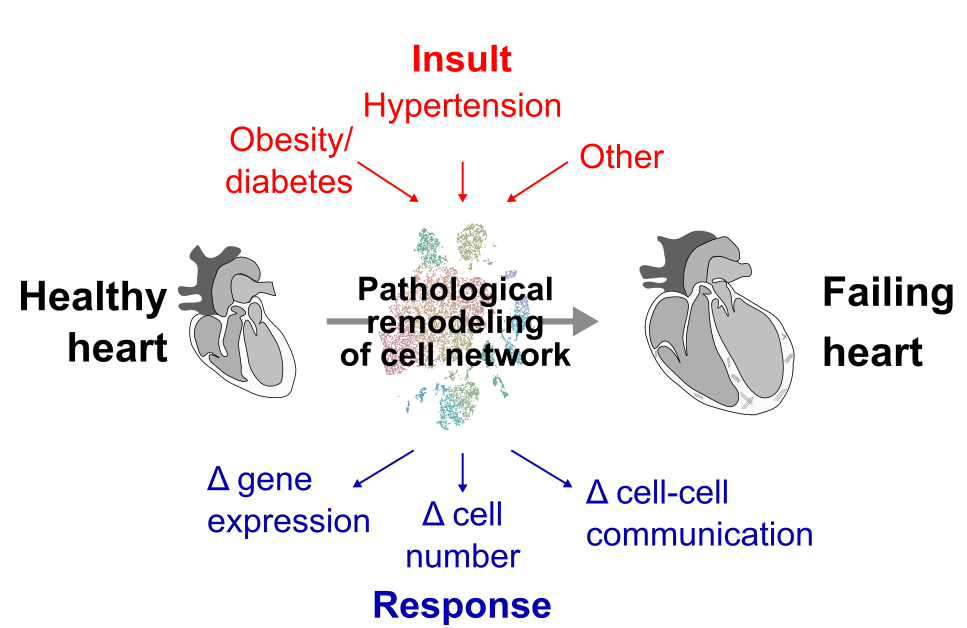
Figure 2: Model of heart failure driven by pathological remodelling of the cardiac cell network elicited by cardiac insults (red) and consequent responses (blue).
Key areas of ongoing research
- Cardiac cell network plasticity and elasticity
Determining how cardiac cell networks respond and adapt to physiological stressors such as obesity and hypertension. - Molecular and cellular drivers
Identifying the molecular and cellular mechanisms that govern these processes. - Novel research tools
Developing unique genetic, computational biology and imaging approaches to precisely study diverse cell populations in the heart.
Key areas of technical expertise
- Single-cell biology
Including single-cell RNA sequencing, high-dimensional flow cytometry and image cytometry. - Multidimensional imaging
Including 3D imaging and spatial mapping of cellular interactions. - Mouse genetics
Including development of novel cell- and organ-specific genetic tools.
Community engagement
We collaborate with the Baker Institute's Community Engagement Group to embed meaningful consumer and community voices into our research. This engagement has helped shape our study direction, refine recruitment and messaging, and support more relevant, accessible and impactful outcomes for people affected by heart failure and cardiac disease.
Key publications
- High-resolution transcriptomic profiling of the heart during chronic stress reveals cellular drivers of cardiac fibrosis and hypertrophy
Circulation 2020 - Cardiac cellularity is dependent upon biological sex and is regulated by gonadal hormones
Cardiovasc. Res. 2020 - New perspectives of the cardiac cellular landscape: mapping cellular mediators of cardiac fibrosis using single-cell transcriptomics
Biochemical Society Transactions 2020

